Visit us at D8 and discover how our regenerative solutions.
Let’s connect at our booth to discuss how Regen Lab is transforming healthcare with innovative, science-driven solutions.
Don’t miss this opportunity to network and learn at one of the premier global congresses in wound care
EWMA 2026 Symposium
Case reports: initial feedback on an autologous innovation in the treatment of chronic wounds
At EWMA 2026, Regen Lab is pleased to invite healthcare professionals to a dedicated symposium focused on innovation in wound care.
This scientific session will present clinical case reports, early feedback from practice, and initial perspectives on an autologous innovation developed to support new approaches in wound care.
Chaired by Mr Gerald Hofer, the symposium will feature presentations from:
Dr Maria Andreea Nagy | Germany
Dr Hester Colboc | France
Through these lectures, attendees will gain insight into:
- early clinical experience with RegenWound-HA Set
- practical case-based observations
- and the opportunities this innovation may offer in the treatment of chronic wounds.
Date: Thursday 7 May 2026
Join us for this important scientific exchange and discover how autologous innovation may help shape the future of chronic wound management.
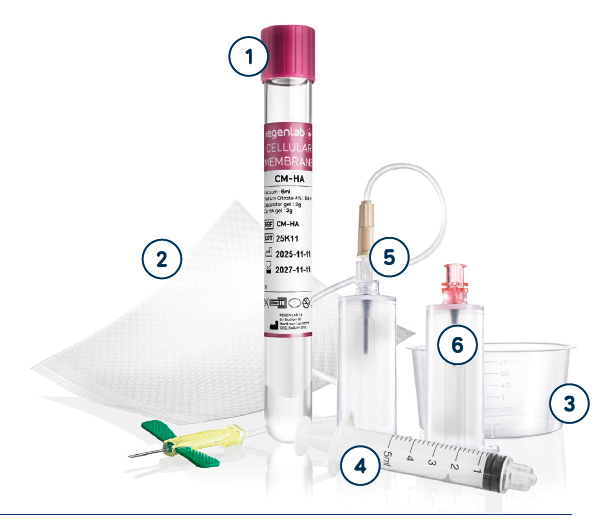
RegenWound-HA Set
MEDICAL PURPOSE
Set for the safe preparation of autologous RegenPRP™ and subsequent wound dressing
COMPOSITION
- Cellular Membrane CM-HA tube
- Silicone Wound Contact Layer
- Surgical bowl
- 5ml syringe Luer-Lock
- Safety-Lok Blood Collection Set with Pre-Attached Holder
- Blood Transfer Device