Qu’est-ce que le RegenKit® Extracell BMC ?
- Système de concentration de moelle osseuse flexible et reproductible
- Dispositif médical de classe IIb certifié CE

PRELEVEMENT DE MOELLE OSSEUSE VOL PAR TUBE11
5 à 10 ml

RÉCUPÉRATION CMN12
> 70%

RÉCUPÉRATION CSM
> 80%
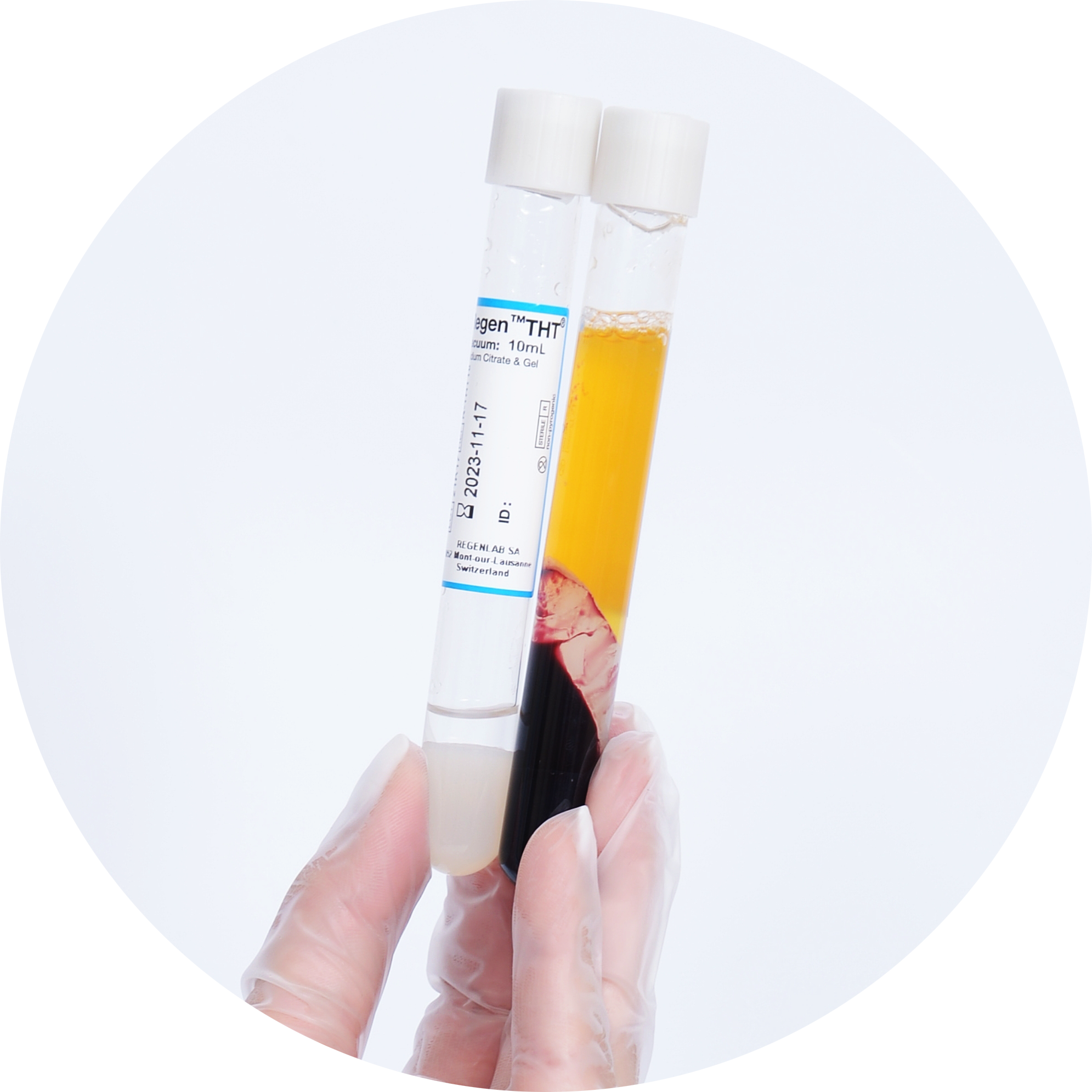
Propriétés des tubes RegenTHT1
Préparation de concentré de moelle osseuse avec RegenKit® Extracell BMC
Le concentré de moelle osseuse (BMC*) est un concentré de cellules autologues provenant d’un prélèvement de moelle osseuse.
La moelle osseuse est un tissu liquide riche d’une population hétérogène de cellules matures et de cellules souches multipotentes (CSM**).
Parmi elles, les cellules stromales mésenchymateuses sont les cellules précurseurs du tissu musculo-squelettique.
Le RegenKit® Extracell BMC est spécifiquement conçu pour obtenir un concentré de moelle osseuse à l’aide d’une centrifugation rapide dans des tubes RegenTHT®. Ces tubes contiennent un gel thixotrope biologiquement inerte d’une densité spécifique qui sépare physiquement le plasma et la fraction de cellules mononucléaires (CMN***) – qui contient les CSM – des autres éléments cellulaires de la moelle osseuse10.
Plus de 80 % des CSM sont récupérées dans le concentré cellulaire.1
In vitro, ces cellules donnent naissance à des colonies de fibroblastes (UFC-F****) pouvant subir une ostéogenèse, une chondrogenèse ou une adipogenèse en fonction du milieu de culture dans lequel elles sont placées.1
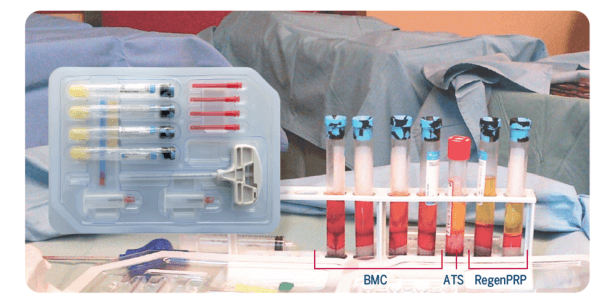
Exemple de protocole :
La moelle osseuse a été prélevée à l’aide des quatre tubes du RegenKit® Extracell BMC. Le BMC résultant peut être combiné avec du plasma riche en plaquettes (RegenPRP) et du sérum de thrombine autologue (ATS), préparés avec un RegenKit® Surgery, pour obtenir un gel.
*BMC = Bone marrow concentrate (concentré de moelle osseuse)
** CSM = Cellules Souches Multipotentes
*** CMN = Cellules mononucléaires
**** UFC-F = Unités de formation de colonies de fibroblastes
Nos produits
Utilisation prévue du dispositif
Préparation d’un concentré de cellules de moelle osseuse autologue
Préparation de plasma riche en plaquettes autologue et d’autres produits dérivés du plasma
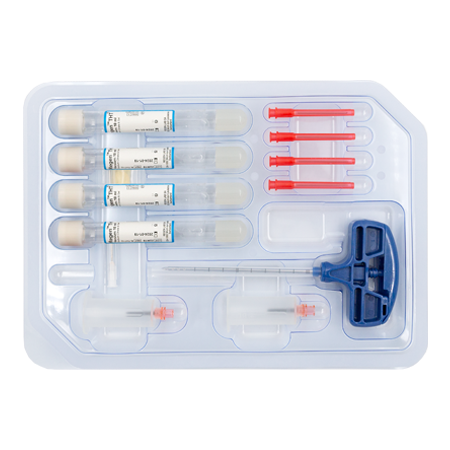

RegenKit® Extracell BMC
1 Trocart
4 Tubes RegenTHT
2 Dispositifs de transfert
4 Aiguilles de transfert
1 Canule de transfert de 80 mm
Pastilles autocollantes
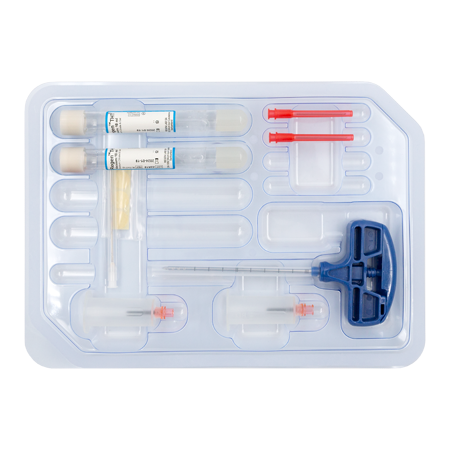

RegenKit® Extracell BMC 2
1 Trocart
2 Tubes RegenTHT
2 Dispositifs de transfert
2 Aiguilles de transfert
1 Canule de transfert de 80 mm
Pastilles autocollantes
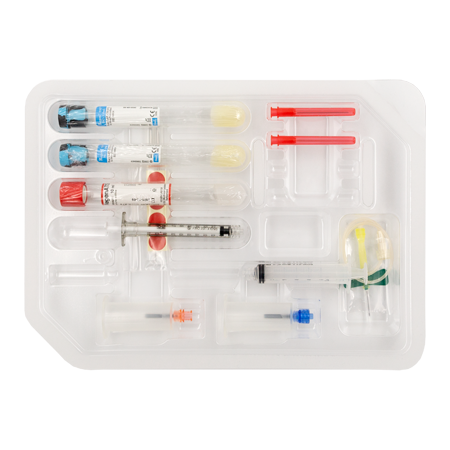

RegenKit® Surgery
1 Aiguille Papillon Safety–LokTM
1 Tulipe de prélèvement
2 Tubes RegenTHT
1 TubeRegenATS
1 Seringue Luer-LokTM de 1ml
1 Seringue Luer-LokTM de 5ml
1 Dispositif de transfert
2 Aiguilles de transfert
2 Canules de transfert de 80 mm
Pastilles autocollantes
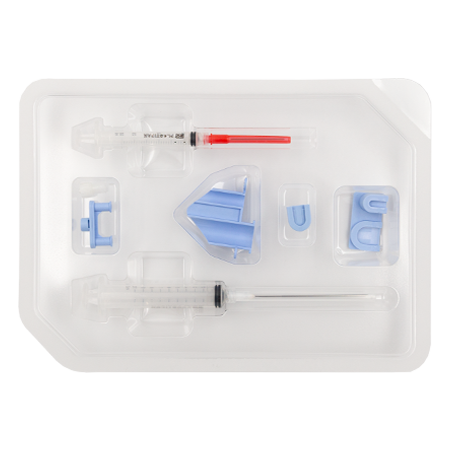

Applicateur RegenSpray
1 Seringue Luer-LokTM de 10 ml
1 Embout pour application en spray
1 Bouchon à double piston
1 Porte-seringue applicateur
1 Seringue Luer-LokTM de 3 ml
1 Adaptateur de rapport 3:10
1 Connecteur Luer-lockTM
1 Canule de transfert de 80 mm
1 Aiguille de transfert
Dispositif médical de classe IIb certifié CE. Regen Lab SA est un fabricant de dispositifs médicaux certifié ISO 13485: 2016 et MDSAP.
Avertissements et précautions
Ce kit est destiné uniquement aux chirurgies de reconstruction osseuse, orthopédiques et traumatologiques.
Une technique d’asepsie stricte doit être suivie pendant toute la procédure. Prendre les précautions de sécurité adaptées pour éviter tout contact avec le sang du patient ou contamination croisée. Prendre les précautions de sécurité adaptées pour se protéger contre les aiguilles ou tubes brisés. Ne pas utiliser les composants stériles de ce kit si celui-ci est ouvert ou endommagé. Ne pas utiliser les composants de ce kit s’ils sont brisés ou comportent un défaut. Ne pas utiliser le tube s’il n’est plus à vide. Ne pas utiliser la solution de citrate de sodium ou d’autres composants du tube séparément. Conserver entre 5 °C et 30 °C ; ramener le kit à température ambiante avant d’utiliser les tubes. Ne pas restériliser, ne pas utiliser après la date d’expiration. Dispositif à usage unique, ne réutiliser aucune pièce du kit. La réutilisation peut entraîner une infection ou d’autres maladies/blessures. L’aiguille de transfert doit être utilisée uniquement pour le transfert de liquides et ne doit pas être utilisée pour l’injection. La préparation du concentré de cellules de moelle osseuse doit être effectuée par un médecin formé à l’équipement et à la procédure, ou sous la supervision du médecin. Le traitement par concentré de cellules de moelle osseuse doit être effectué par un médecin qualifié. Ne pas injecter de concentré de cellules de moelle osseuse en intravasculaire. Le patient doit être informé des risques généraux associés au traitement et des effets indésirables possibles. L’innocuité et l’efficacité de l’association du concentré de cellules de moelle osseuse à d’autres thérapies doivent être évaluées par le médecin. L’innocuité et l’efficacité n’ont pas été évaluées chez les enfants et chez les femmes enceintes ou allaitantes.
Le concentré de cellules de moelle osseuse doit être préparé à partir de sang frais et doit être utilisé dans les quatre heures (usage extemporané uniquement). Tous les tubes et composants du kit doivent être entièrement mis au rebut selon la méthode d’élimination après chaque utilisation afin d’éviter toute contamination potentielle avec les produits sanguins. Utiliser une centrifugeuse avec rotor à angle fixe de 45° ou une centrifugeuse horizontale à godet oscillant (ex. RegenPRP Centri fournie par Regen Lab). Suivre les instructions du fabricant lors de l’utilisation de la centrifugeuse. Les tubes doivent être centrifugés, comme recommandé dans le manuel d’utilisation, à une force de centrifugation relative (RCF) de 1500 g. Une RCF excessive (supérieure à 2200 g) peut entraîner une rupture du tube et donc une exposition au sang ainsi que d’éventuelles blessures. Une RCF inférieure à 1500 g peut entraîner une séparation incorrecte du sang et une contamination du concentré de cellules de moelle osseuse par les érythrocytes. La taille des supports et inserts de centrifugation doit être adaptée aux tubes. L’utilisation de supports trop grands ou trop petits peut entraîner la rupture des tubes. Veiller à ce que les tubes soient correctement placés dans les supports de centrifugation. Les tubes doivent être équilibrés dans la centrifugeuse
Effets secondaires possibles
Hémorragie, hématome, cicatrisation retardée de la plaie. Infection opératoire précoce ou tardive. Douleur persistante, séromes. Réaction allergique à l’anesthésie.
Breveté par Regen Lab SA
Platelet Rich Plasma
Brevet U.S. US8529957, US11241458, US11110128, US11096966, US10881691,
US10092598, US10080770, US10064894, US8529957, US9833478, US10052349, US11241458
Brevet Européen EP2073862B, EP3111974B1
Brevet Suisse CH696752
Brevet Hong Kong HK1231793
Bibliographie
- Barry, F., A Report on The Phenotypic Characterisation of Mesenchymal Stem Cells isolated from Human Bone Marrow Using the RegenTHT, A-CP and Regen BCT Blood Separation Systems. 2015, The Regenerative Medicine Institute (REMEDI), National University of Ireland Galway.
Contacter Regenlab
Changez la vie de vos patients en leur proposant un traitement adapté, simple et sûr.
Pour plus d'informations sur nos produits, merci de nous contacter en cliquant sur le bouton ci-dessous :
Nos bureaux régionaux sont situés à :
- New York (USA)
- Montréal (Canada)
- Venise (Italie)
- Munich (Allemagne)
- Paris (France)
- Dubaï (U.A.E.)
- Beijing (Chine)
- Istanbul (Turquie)




